|
Then, if any of these levels fall outside of a preset range, a notification is sent to the base station, smartphone, or tablet. Why? Well, the Owlet sock tracks a baby’s oxygen levels and heart rate while he or she sleeps. 
The Owlet Smart Sock 2 promises parents of newborns something that they haven’t felt since they found out they were pregnant-peace of mind. The company states 94% of parents who use the smart sock report improvements in their own sleep, as well as reduced anxiety. Instead, check out our picks for best baby monitors of 2022 + full list of baby monitor reviews over here. Without further research and testing, we are not recommending Owlet products. It presents our opinions on Owlet at the time, but should be read only with the above context. The below review was published in 2020 prior to these events. That’s a big change and a rather concerning turn of events for Owlet. 
It’s the same sock, but focused now on monitor sleep quality, not vitals like blood oxygen level and heart rate. Since then, Owlet has changed the Smart Sock to the Dream Sock. It’s worth noting this warning had to do with the company’s marketing, not the actual performance of this product, but it’s still a big asterisk on Owlet. "We're all signing the petition with one goal in mind, to save the Smart Sock so it can one day save another parent from a headache or lack of sanity," Smith said.IMPORTANT NOTE: in late 2021, Owlet received a warning letter from the FDA regarding the misclassification of its Smart Sock as a medical device. When questioned on whether customers can continue using the Smart Socks, the company said "the product’s functionality has not changed at this time. We will notify customers of any updates to the Smart Sock products that have already been distributed." Owlet pointed out the FDA didn't “identify any safety concerns about the Smart Sock,” and claimed the safety of the socks had "been validated by third parties, in which it was shown to be safe." In response to the FDA, Owlet posted their own letter on its site, explaining they plan to comply with the agency's request and seek marketing approval going forward. Since 2016, the FDA has contacted Owlet about altering their marketing of the baby monitoring socks, according to the letter. 10/10 would recommend it □- kp November 28, 2021 
So glad me and my bf got the Owlet Smart Sock definitely a must have for a newborn! We’re able to sleep through the night knowing our baby is ok. Kelli Stout signed the petition and commented the device lead doctors to discovering her child had sleep apnea. It's the only way I can sleep and know my child is okay," Smith said. "The FDA is going after the device, the Smart Sock is so necessary for parents now. Marny Smith, who also signed the petition, is a mother of three and told USA TODAY without the Smart Sock she'd lose her "mental health" while nursing and caring for her children. This is a product that has given peace of mind to millions of parents," Needham said in the petition. "I can’t say for sure what would have happened had the monitor not gone off but I’m thankful it did. She stayed asleep until her she heard her daughter's Smart Sock alert go off and realized her child was pressed against her side and underneath her breast. One night while Needham was nursing her newborn, she fell asleep. Use your voice! - Sign the Petition! via Zeke November 24, 2021 This smart sock literally saved our baby’s life by alerting us to low oxygen levels in the middle of the night.įight the FDA-Keep the Owlet Smart Sock on the market. Have you checked your chicken?: Nearly 98,000 pounds of raw chicken patty products recalled after reports of bone pieces Hand sanitizer recall: Hand sanitizer recalled because packaging looks like water bottles, company says 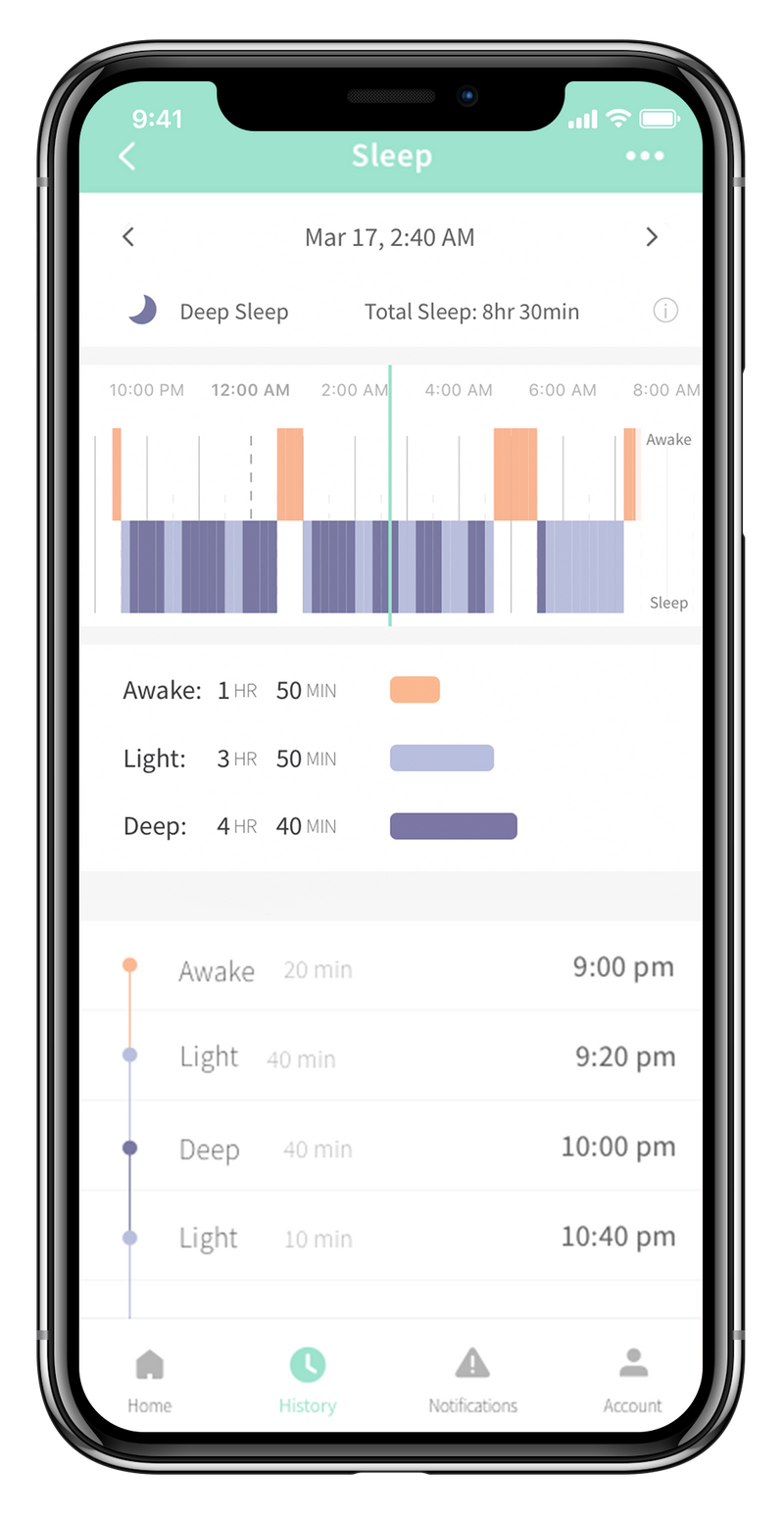
The petition has garnered over 150,000 signatures of concerned parents who say the Smart Socks give them "peace of mind." "Keeping this product on the market would ensure peace of mind for millions of parents and quite literally save lives by alerting parents to an issue before it’s too late," Caitlin Needham said in her petition to keep the Smart Sock. The FDA stated the socks were medical devices since they provide heart rate and oxygen levels but Owlet claimed the smart socks were "low-risk products." In the warning letter, the FDA said the company has sold the product without proper “marketing approval, clearance, or authorization” from the FDA.Īs of Tuesday morning, the Owlet Sock family of products is "currently unavailable,” according to the Smart Socks’ product page, and thousands of parents are unhappy. Owlet’s Smart Socks, baby monitoring socks programmed to track a baby’s sleep patterns, have halted sales after a warning letter from the U.S.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
